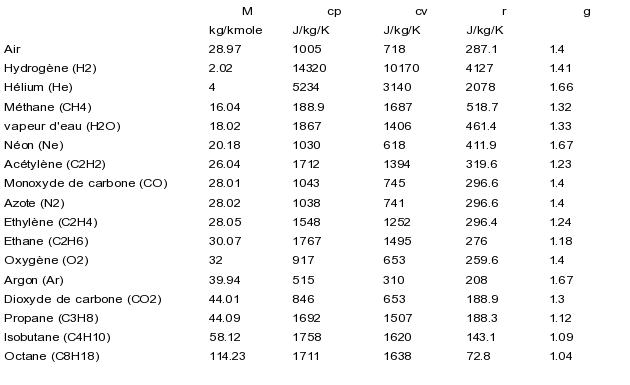
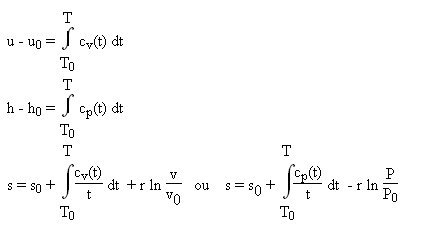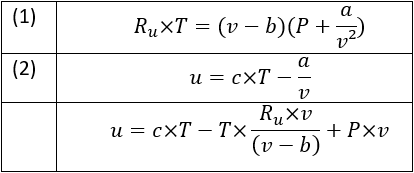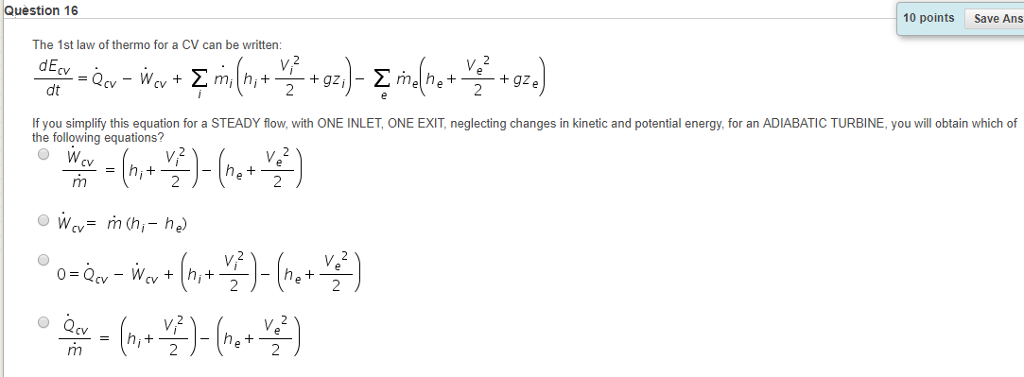Use the thermodynamic relations to show that for an ideal gas CP − CV = R. - Sarthaks eConnect | Largest Online Education Community

Thermo Scientific™ Dri-Cal™ Particle Size Standards 5.3μm ± 0.3μm, σ = 0.6μm, 11% CV Thermo Scientific™ Dri-Cal™ Particle Size Standards | Fisher Scientific

IIT/JEE Relation In Cp & Cv (Heat Capacity at Constant Pressure & Volume). Thermo Chemistry(Part-38) - EduGorilla Trends - Videos, News, Career Updates

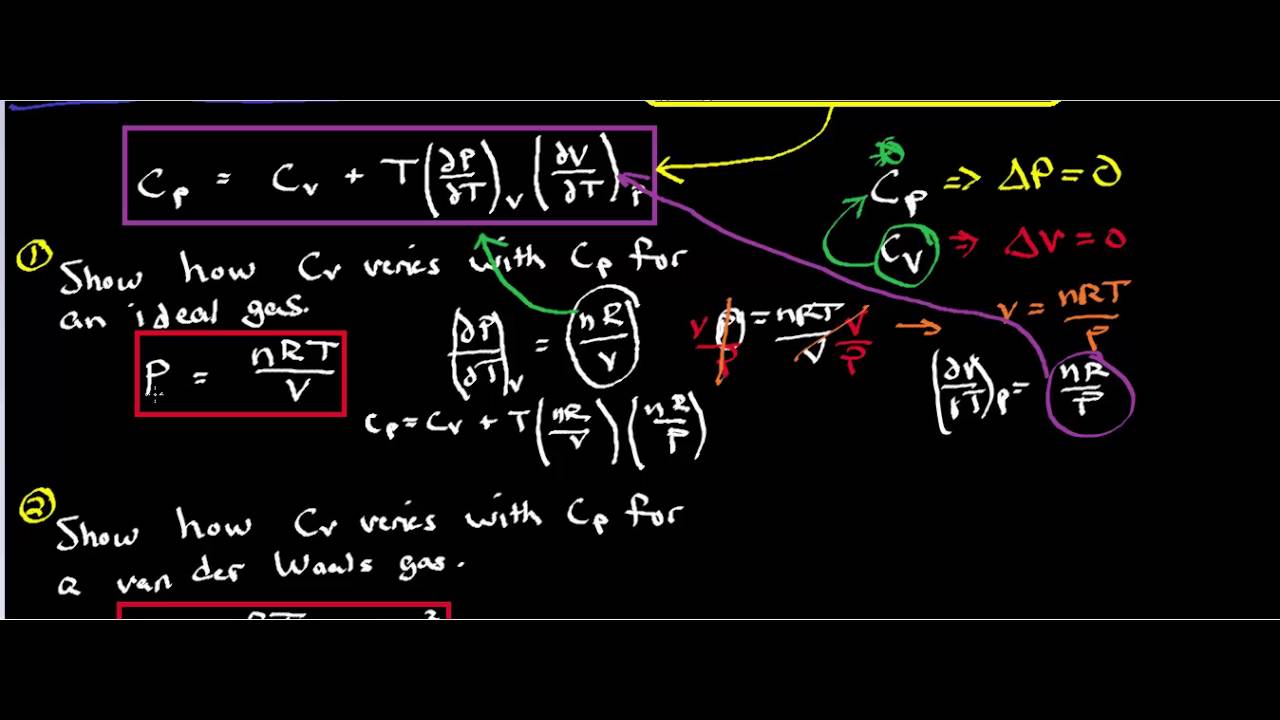
SOLVED: By using the second law of thermodynamics and the definitions of U, HAG, Cp, Cv, the expansion coefficient, and the isothermal compressibility K, and using calculus, prove that: aCp da =

Kinesis CV-TSP-206 Inlet Check Valve, Thermo Scientific 8100, 8700; 1/EA from Cole-Parmer United Kingdom

SOLVED: 5. A general relation between Cp and Cy. (a) Using the thermodynamic identity, show that the heat capacity at a fixed volume V is expressed as: Cv = T (∂S/∂T)V (b)