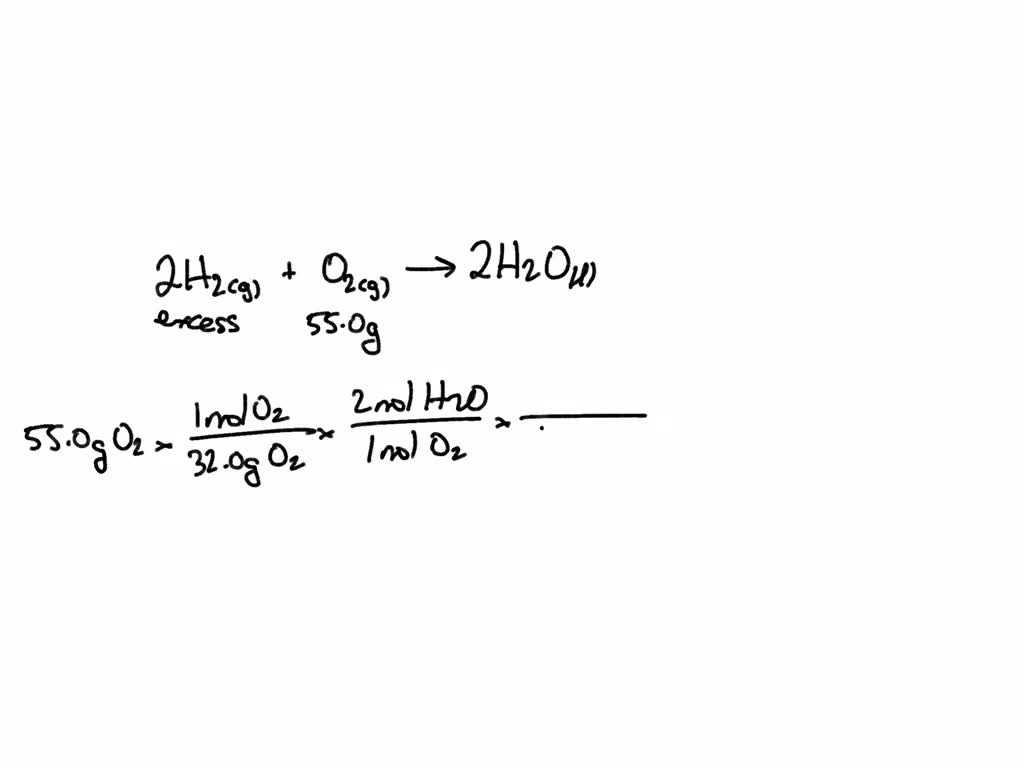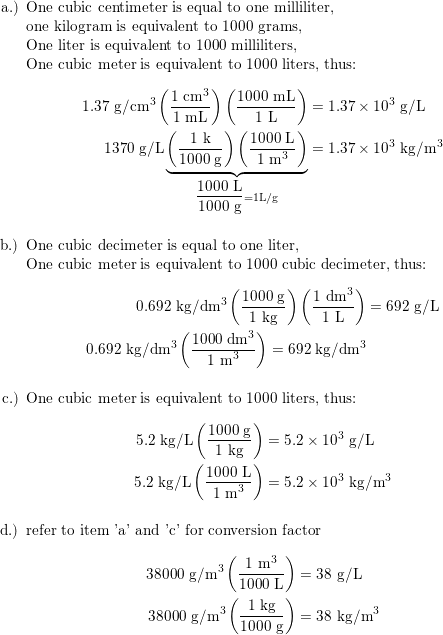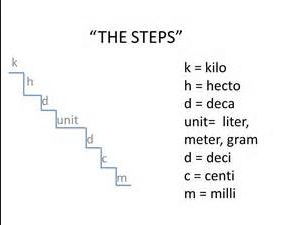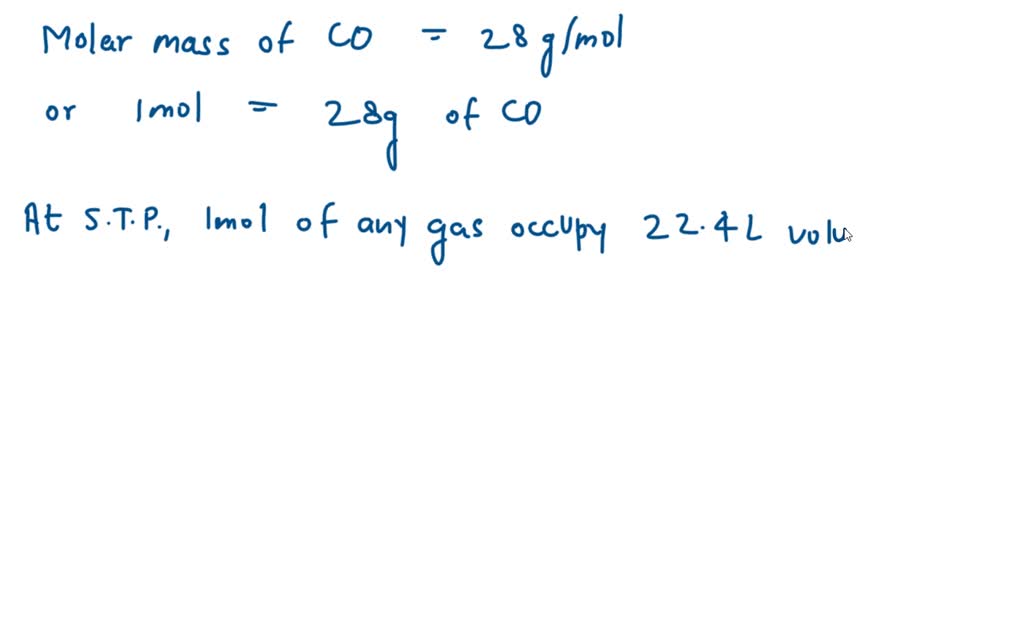
SOLVED: What is the mass in grams of 1 liter of carbon monoxide, CO, at standard temperature and pressure, STP? Note the molecular weight of CO is 28 g/mole and at STP,

Meters, Liters, and Grams: Understanding the Metric System: Kay Nation, Lynn Sweat: 9780801549526: Amazon.com: Books

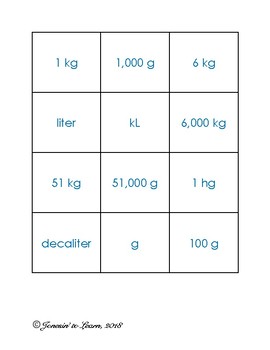
metric meterlitergramwkst copy.pdf - Name Date Per Writing Meters Liters and Grams When Moving from one unit to a larger unit No. of units moved Unit | Course Hero
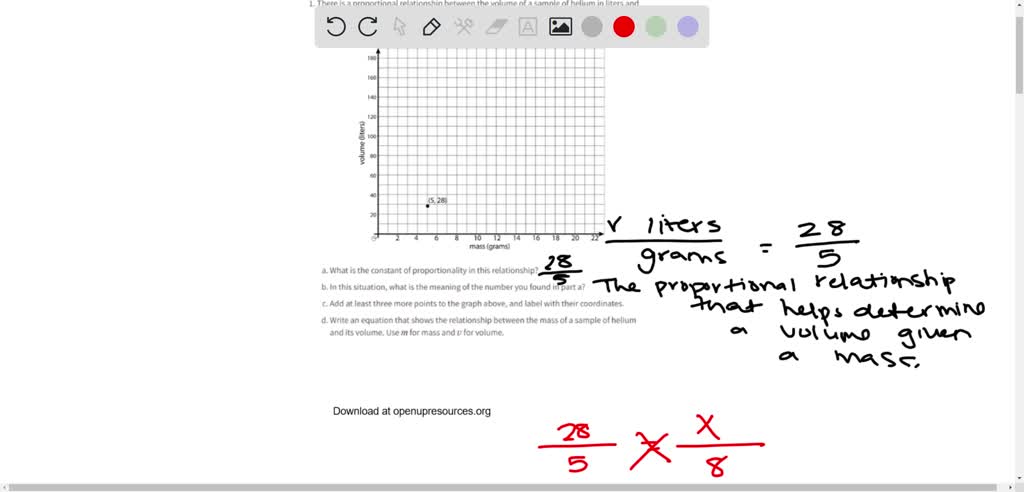
SOLVED:There is a proportional relationship between the volume of a sample of helium in liters and the mass of that sample in grams. If the mass of a sample is 5 grams,

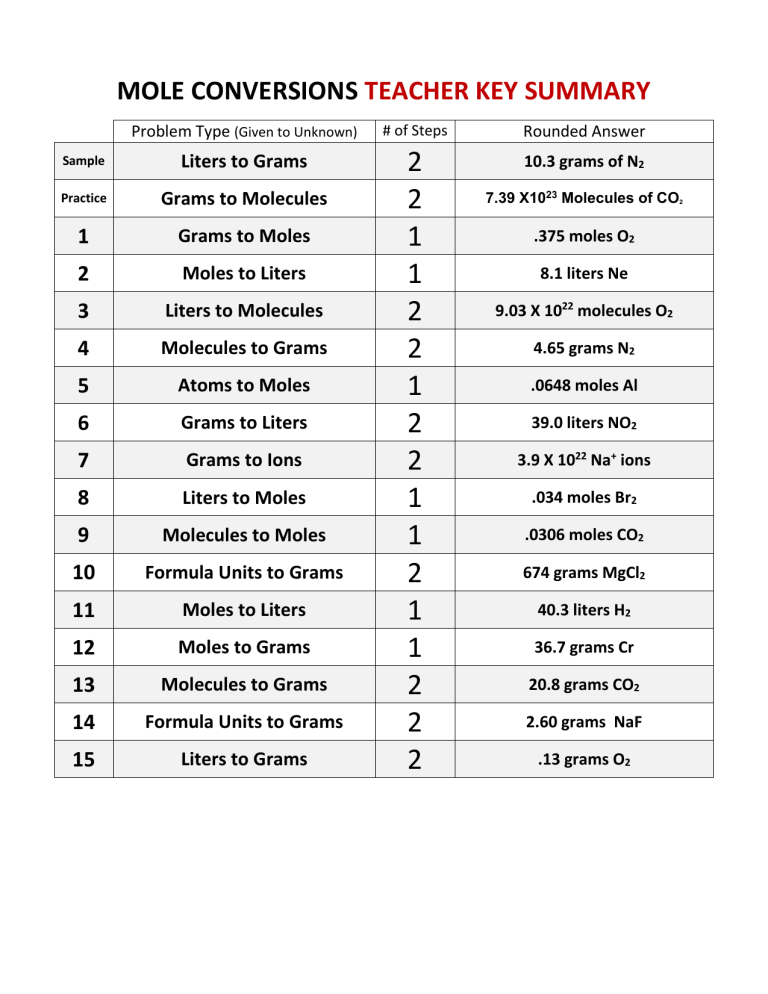
Intro to Quantities Review Problems These are the calculations you should be able to perform: Sum of molar mass for a compound Convert mass mole Convert. - ppt download